Viral RNA & DNA Extraction


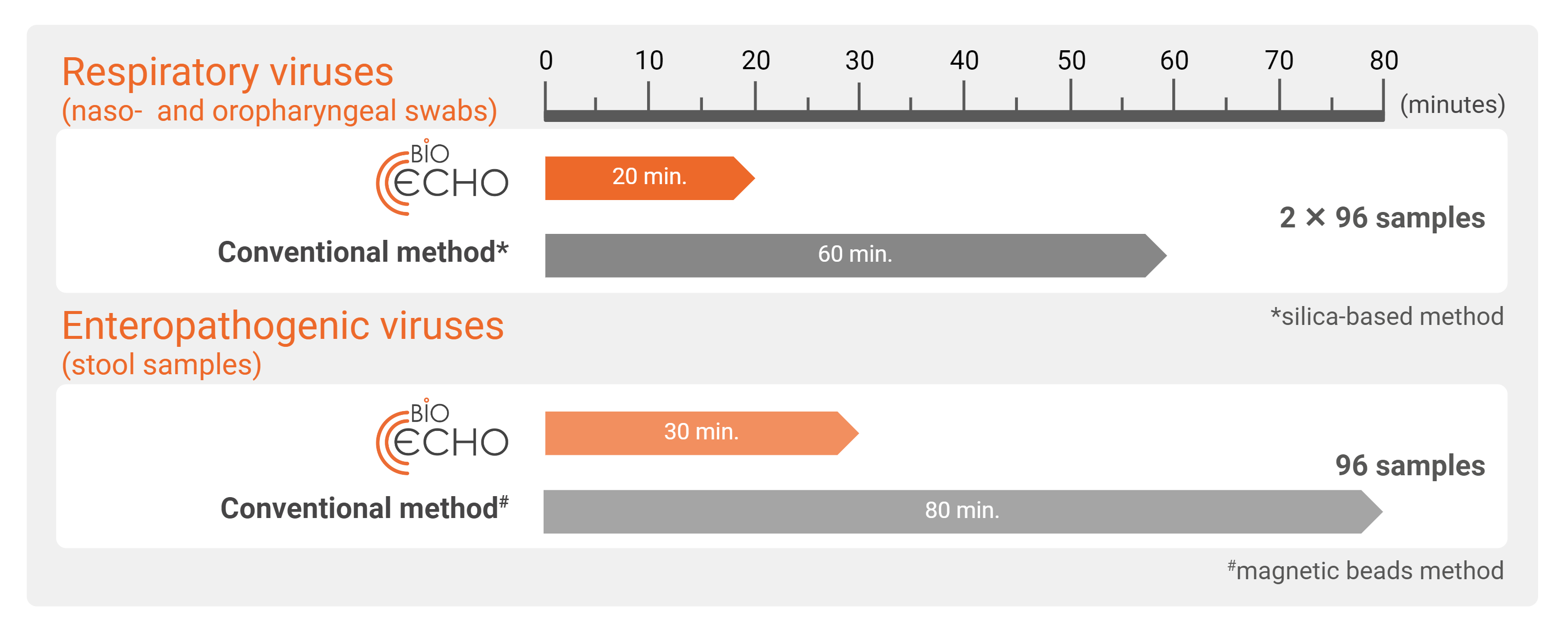
3 x faster
Compared to conventional methods

~70% fewer handling steps
Compared to silica-based methods

Inhibitor-free RNA and DNA
For superior downstream performance


“Our research facility is active in phase I and phase II clinical trials with our lab performing the safety and screening assays. When I heard about the EchoLUTION Viral RNA/DNA Kit, I was curious to see how it works and how it could possibly be integrated into our workflow. Testing it, I was surprised by its simplicity and how clean and fast the protocol was. The main competitor kits on the market have a lot more steps and a bigger footprint. And finally, when I heard about the sustainability concept of BioEcho, I was definitely convinced!”
Kirk Lum CGMBS,
Associate Director, AltaSciences, Cypress, CA.

“Our diagnostic lab is using the EchoLUTION Viral RNA/DNA Swab Kit to serve the high testing demands during the pandemic. The technology allows us to extract nucleic acids fast and efficiently. Also, I would like to highlight the excellent collaboration we have with BioEcho: Even in these stressful times, their employees are very engaged and supportive.”
Dr. Bernhard Zöllner,
Medical Director, Medical Care Center (MVZ), Bioscientia Healthcare GmbH, Moers, Germany

“Fast results are essential for patient management to avoid outbreaks. With the EchoLUTION Viral RNA/DNA Swab Kit from BioEcho in combination with a fast PCR assay, we can process a large number of samples in a short time. Standard laboratory equipment can be used for processing. This makes it easy to establish the method in the laboratory.”
Dr. Thomas Brodegger,
MEDILYS Laborgesellschaft mbH, human medical diagnostics laboratory, Hamburg, Germany